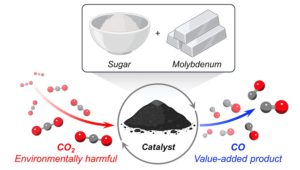
Molybdenum-sugar-based catalyst effectively destroys CO2

A study by Northwestern University researchers has found that a catalyst made from earth-abundant molybdenum and common table sugar has the power to destroy carbon dioxide (CO2) gas.
To come up with this solution, the first thing the scientists had to do was transform molybdenum into molybdenum carbide using a carbon source. They discovered a cheap option in an unexpected place: the pantry. Surprisingly, sugar—the white, granulated kind found in nearly every household—served as an inexpensive, convenient source of carbon atoms.
Once this was done, the team was able to use the catalyst to transform CO2 into carbon monoxide (CO), an important building block to produce a variety of useful chemicals. When the reaction occurs in the presence of hydrogen, for example, CO2 and hydrogen transform into synthesis gas (or syngas), a highly valuable precursor to producing fuels that can potentially replace gasoline.
Operating at ambient pressures and high temperatures (300–600 degrees Celsius), the catalyst converted CO2 into CO with 100% selectivity.
High selectivity means that the catalyst acted only on the CO2 without disrupting surrounding materials. In other words, industry could apply the catalyst to large volumes of captured gases and selectively target only the CO2. The catalyst also remained stable over time, in other words, it stayed active and did not degrade.
“In chemistry, it’s not uncommon for a catalyst to lose its selectivity after a few hours,” Omar K. Farha, the study’s senior author, said in a media statement. “But after 500 hours in harsh conditions, its selectivity did not change.”
This is remarkable because CO2 is a stable—and stubborn—molecule.
“Converting CO2 is not easy,” Milad Khoshooei, co-lead author of the study, said. “CO2 is a chemically stable molecule, and we had to overcome that stability, which takes a lot of energy.”
Developing materials for carbon capture is a major focus of Farha’s laboratory. His group develops metal-organic frameworks (MOFs), a type of highly porous, nano-sized materials that are like “sophisticated and programmable bath sponges.” Farha explores MOFs for diverse applications, including pulling CO2 directly from the air.
In the researcher’s view, MOFs and the new catalyst could work together to play a role in carbon capture and sequestration.
“At some point, we could employ a MOF to capture CO2, followed by a catalyst converting it into something more beneficial,” Farha suggested. “A tandem system utilizing two distinct materials for two sequential steps could be the way forward.”
“This could help us answer the question: ‘What do we do with captured CO2?’,” Khoshooei said. “Right now, the plan is to sequester it underground. But underground reservoirs must meet many requirements in order to safely and permanently store CO2. We wanted to design a more universal solution that can be used anywhere while adding economic value.”
{{ commodity.name }}
{{ post.title }}
{{ post.date }}

Comments
Ken Towe
The point of carbon capture is to lower atmospheric CO2 levels enough to affect the Earth’s climate. Given that just ONE ppm of CO2 equals 7.8 gigatons any solution must convert and store massive amounts (billions of tons) to have any noticeable effect. And, the process must be carbon-neutral itself. This makes almost any solution unworkable from a cost-benefit point of view.