Scientists develop low-cost, graphene-based method to remove uranium from drinking water

A research team at the Massachusetts Institute of Technology has developed a highly efficient method for removing uranium from drinking water.
In a paper published in the journal Advanced Materials the MIT group together with colleagues from the Argonne National Laboratory, the University of Tokyo and National Chiao Tung University, tested the idea of applying an electric charge to graphene oxide foam to capture uranium in solution, which precipitates out as a condensed solid crystal.
“Within hours, our process can purify a large quantity of drinking water below the EPA limit for uranium,” Ju Li, one of the study co-authors, said in a media statement.
Earlier research has shown that electrically charged carbon fibre could filter uranium from water, however, results from those tests were partial and imprecise.
But after investigating the behaviour of graphene foam used for lithium-sulphur batteries, Li and her team found that the physical performance of this foam was unique because it has the ability to attract certain chemical species to its surface.
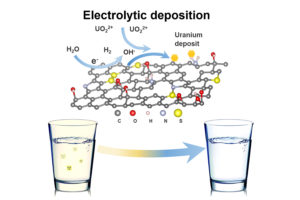
The work basically consisted of transforming graphene foam into the equivalent of a uranium magnet. By sending an electric charge through the foam, splitting water and releasing hydrogen, the researchers were able to increase the local pH and induce a chemical change that pulled uranium ions out of the solution.
They then found that the uranium would graft itself onto the foam’s surface, where it formed a never-before-seen crystalline uranium hydroxide. On reversal of the electric charge, the mineral, which resembles fish scales, slipped easily off the foam.
According to Li, every time the uranium filtration process is used, the foam can capture four times its own weight of uranium and can achieve an extraction capacity of 4,000 mg per gram, which is a major improvement over other methods.
“We’ve also made a major breakthrough in reusability because the foam can go through seven cycles without losing its extraction efficiency,” Li said.
The graphene foam functions as well in seawater, where it reduces uranium concentrations from 3 parts per million to 19.9 ppb, showing that other ions in the brine do not interfere with filtration.
Given these results, the team believes its low-cost, effective device could become a new kind of home water filter, fitting on faucets like those of commercial brands.
“Some of these filters already have activated carbon, so maybe we could modify these, add low-voltage electricity to filter uranium,” Li said.
Besides looking into commercial applications, the researchers are also investigating the possibility of modifying the filter to be selective for other heavy metals such as lead, mercury, and cadmium.
The inspiration
Before focusing on developing the filter, Li and his colleagues were looking to find better approaches to environmental cleanup of heavy metals from mining sites.
However, after reviewing documentation from the U.S. Geological Service and the Environmental Protection Agency, the scientists realized that taking a deeper look at uranium water contamination was urgent.
They found that there are unhealthy levels of the radioactive metal moving into reservoirs and aquifers from natural rock sources in the northeastern United States, from ponds and pits storing old nuclear weapons and fuel in places like Hanford, Washington, and from mining activities in many western states.
A concrete example is that of High Plains and Central Valley aquifers, which supply drinking water to 6 million people and which, together with other sites, show uranium concentrations close to or above the EPA’s recommended ceiling of 30 parts per billion — a level linked to kidney damage, cancer risk, and neurobehavioral changes in humans.
{{ commodity.name }}
{{ post.title }}
{{ post.date }}

Comments