‘Ultimate’ anode for next-generation batteries to become more efficient

A recent study by researchers at Korea’s Daegu Gyeongbuk Institute of Science and Technology found that high concentration electrolyte (HCE) significantly improves Li+ ion transport at lower temperatures and thermal stability of solid-electrolyte interface at higher temperatures, thereby increasing the cycling performance of lithium metal batteries (LMBs).
In the article published in the Chemical Engineering Journal, the scientists explain that, compared to lithium-ion batteries, LMBs have a very high energy density and charge very quickly. Yet, they suffer from low current efficiency, poor cyclability and are prone to li-dendrite formation— or excess lithium deposition on the electrodes.
So far, research has found that using high salt concentration electrolytes (HCE) diluted with ether-based solvents can solve these problems and improve performance. However, a proper understanding of how HCE dilution affects the working of LMBs over a wide range of working temperatures is still a lingering question.
This is where the DGIST experts come in. After exploring the effect of HCE dilution on lithium metal batteries’ cycle over a wide range of temperatures, the paper’s senior authors, Hongkyung Lee and Hochun Lee concluded that HCEs are often vicious and that diluting them can increase the ion migration within the cell and improve wetting of electrodes.
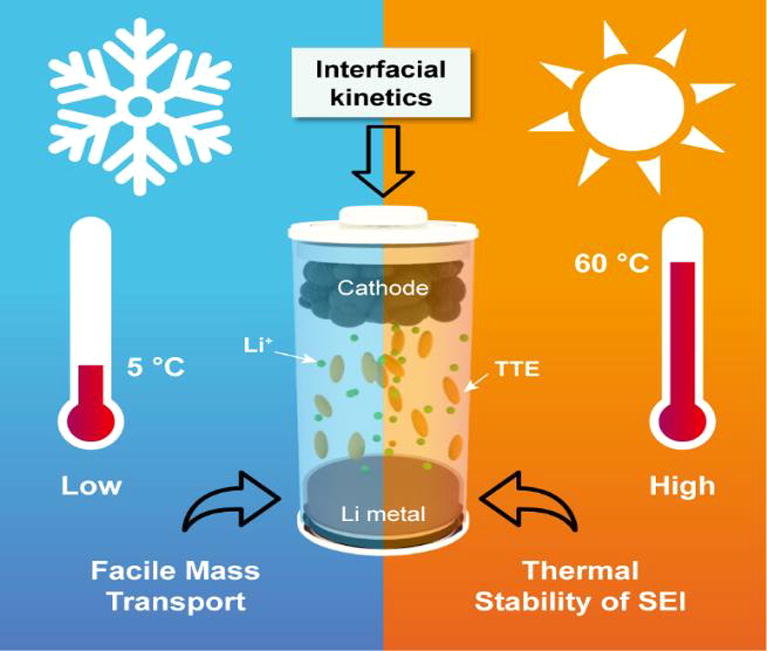
The team adopted a new HCE dilution technique that allowed them to demonstrate good LMB cycling performance at temperatures between 2–60°C. They then conducted a comparative electrochemical analysis of a model HCE well as an HCE diluted with 1,1,2,2-tetrafluoroethyl 2,2,3,3-tetrafluoropropyl ether (TTE).
The experimental results indicated that TTE dilution significantly improved Li+ ion transport and reduced dendritic Li plating at low temperatures, which is essential for maintaining cycling stability.
TTE was also found to be responsible for the formation of the thermally stable solid-electrolyte interface that determines the high-temperature cycling ability of LMBs. The analysis also revealed that TTE dilution could also prove beneficial for the high voltage cycling of Li cells.
“The electrolyte-deterministic interfacial stability is a crucial concern for securing battery performances,” Hongkyung Lee said in a media statement. “This work provides a rational strategy for diluting high-concentration electrolytes to stabilize a highly reactive Li surface. The findings in this study can offer the clues to design electrolyte microstructure, identify its fundamental impact on the interfacial stability over a wide temperature range, and contribute toward stable cycling of Li-metal batteries in practice.”
In Lee’s view, the lithium-metal is considered as an ultimate anode for next-generation batteries, which means that the insights from this study can be applied to design small and light but efficient devices with long-term cycling stability that can act as power sources for drones, walking robots and physical augmentation mechanisms, among other things.
“With our research, we tried to reinforce the development of longer-cycling, higher-energy density batteries without sacrificing the charging rate, which is a prerequisite of electric vehicles that have longer mileage,” Lee said.
{{ commodity.name }}
{{ post.title }}
{{ post.date }}

Comments