New sensor able to detect rare earths in acid mine waste

Penn State University researchers have developed a luminescent sensor that can detect terbium, one of the rarest of the rare earth elements, in complex environmental samples like acid mine waste.
In a peer-reviewed article published in the Journal of the American Chemical Society, the scientists explain that the sensor takes advantage of a protein that very specifically binds to rare earth elements. This means that the device could be harnessed to help develop a US supply of these metals.
According to the researchers, even though there is not currently a domestic supply chain of rare earth elements like terbium, they are quite abundant in non-traditional sources such as coal byproducts, acid mine drainage, and electronic waste. However, to develop these sources of REE, robust detection methods are required and, until now, the gold standard was to use a type of mass spectrometry called ICP-MS which is expensive and not portable. Portable methods, on the other hand, are not as sensitive and do not perform well in complex environmental samples, where acidic conditions and other metals can interfere with detection.
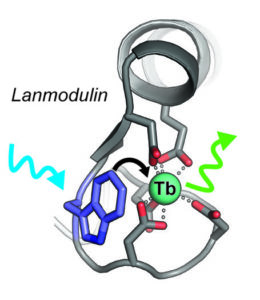
Unlike these methods, Penn State’s development relies on lanmodulin, a protein that is almost a billion times better at binding to rare earth elements than to other metals. The protein’s selectivity to bind rare earth elements is ideal for a sensor, as it is most likely to bind to rare earths instead of other metals that are common in environmental samples.
To optimize lanmodulin as a sensor for terbium specifically, the researchers altered the protein by adding the amino acid tryptophan to the protein.
“Tryptophan is what is called a ‘sensitizer’ for terbium, which means that light absorbed by tryptophan can be passed to the terbium, which the terbium then emits at a different wavelength,” Joseph Cotruvo Jr., senior author of the study, said in a media statement.
“The green color of this emission is actually one of the main reasons terbium is used in technologies like smartphone displays. For our purposes, when the tryptophan-lanmodulin compound binds to terbium, we can observe the emitted light, or luminescence, to measure the concentration of terbium in the sample.”
The researchers developed many variants of the tryptophan-lanmodulin sensor, optimizing the location of the tryptophan so that it does not interfere with lanmodulin’s ability to bind to rare earth elements. These variants provided important insights into the key features of the protein that enable it to bind rare earths with such high selectivity.
Then, they tested the most promising variant to determine the lowest concentration of terbium the sensor could detect in idealized conditions — with no other metals to interfere. Even under highly acidic conditions, the sensor could detect environmentally relevant levels of terbium.
“One challenge with extracting rare earth elements is that you have to get them out of the rock,” Cotruvo said. “With acid mine drainage, nature has already done that for us, but looking for the rare earths is like finding a needle in a haystack. We have existing infrastructure to treat acid mine drainage sites at both active and inactive mines to mitigate their environmental impact. If we can identify the sites with the most valuable rare earth elements using sensors, we can better focus extraction efforts to turn waste streams into revenue sources.”
Next, the researchers tested the sensor in actual samples from an acid mine drainage treatment facility in Pennsylvania — an acidic sample with many other metals present and very low levels of terbium — 3 parts per billion. The sensor determined a concentration of terbium in the sample that was comparable to what they detected with the ‘gold standard’ method, suggesting that the new sensor is a viable way to detect low concentrations of terbium in complex environmental samples.
“We plan to further optimize the sensor so that it is even more sensitive and can be used more easily,” Cotruvo said. “We also hope to target other specific rare earth elements with this approach.”
{{ commodity.name }}
{{ post.title }}
{{ post.date }}

Comments